Traumatic brain injury (TBI) from blunt impact or explosives is associated with vision symptoms such as vision loss, light sensitivity and headaches, including migraines.
These aftereffects are commonly clumped together with other symptoms in patients who have had a jarring and bruising insult to the brain. In short, the visual effects have been thought to be due to TBI.
At Vanderbilt Eye Institute, visual system neuroscientist Tonia Rex, Ph.D., is using her research as an argument against such generalizations, specifically in victims of detonated improvised explosive devices (IEDs) and similar blasts. Rex has talked with veterans of the Gulf wars for more than a decade and designed studies to help treat their eye injuries. In doing so, she has demonstrated that the damage causing these ocular symptoms can originate from the retina and optic nerve, and not just the brain.
Working in collaboration with David J. Calkins, Ph.D., assistant vice president for research and director of the Vanderbilt Vision Research Center, and Craig Duvall, Ph.D., a professor of biomedical engineering, Rex used a mouse model to test the microparticle delivery of erythropoietin (EPO-R76E), a protective cytokine. The delivery method can provide slow release of this neuroprotective antioxidant near the eye.
“The results of our work confirm the neuroprotective effects of EPO-R76E,” Rex said. “Our erythropoietin microparticles are a promising carrier worthy of further development for sustained therapeutic delivery in models of neurodegeneration.”
The Eye as Gateway
In earlier work, Rex showed that the repeat insult of explosives such as IEDs triggers a very different phenomenon than the TBI typically suffered by football players, for example.
“The IEDs that our fighting men and women are exposed to so often is not really a single blast event,” Rex said. “The impact of these explosives rebounds off surfaces such as walls and the ground, with the net effect of repeated pressure assaults on the soldier. Even in lower-level hits, this reverberation has an exponential effect on injuries.”
Through her research, she has shown that these rebounding blasts have the potential to damage the interior pathways of vision, the retinal ganglion cells and the optic nerve. Understanding the eye as a gateway for injury, she and Duvall designed the microparticle delivery system to help mitigate or reverse damage in retinal ganglion cells and ocular structures.
“These slow-release microcapsules resulted in significant reduction in inflammation and axon degeneration and preservation of visual function.”
The Erythropoietin Microparticle
The researchers used erythropoietin, a cytokine protein that increases levels of antioxidant proteins and packaged them within microspheres that are reactive oxygen species scavengers.
“Recently, microparticle-mediated delivery of erythropoietin for nerve regeneration has gained interest,” Rex said. “We and others have previously demonstrated that erythropoietin can preserve retinal neurons and optic nerve axons in a variety of models of neuronal cell death, including our model of blast-induced indirect optical neuropathy.”
In Vitro and Murine Testing
Rex and Duvall tested this delivery mechanism through an in vitro oxidative stress assay and a mouse model of blast-induced indirect traumatic optic neuropathy.
“Craig Duvall invented the biochemical ‘skin’ of these microparticles, and I have been working on the therapeutic applications for them,” she said.
They delivered microspheres of EPO-R76E by injection into the eye near the retina, where reactive oxygen species are present.
“We found that these slow-release microcapsules resulted in significant reduction in inflammation and axon degeneration and preservation of visual function as measured by visual evoked potentials,” Rex said.
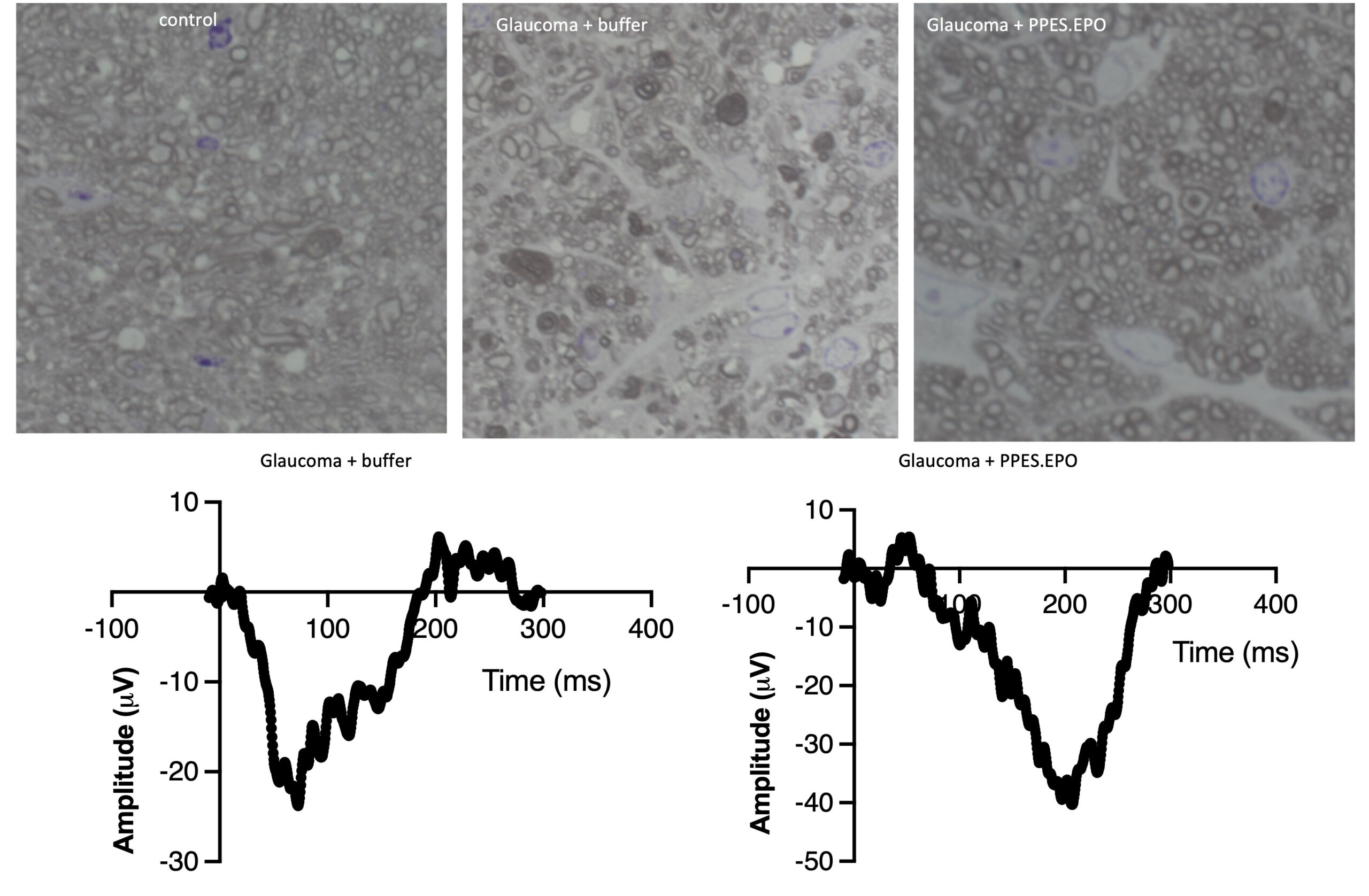
Erythropoietin Magic
Now, Rex and her colleagues are testing efficacy of this approach in models of glaucoma with the hope of moving toward human trials and to integration of the therapy into treatment regimens for a wider range of ophthalmological patients.
Erythropoietin also has applications beyond neurotrauma or other diseases of the eye, just not yet in a deliverable form, Rex said.
“Our work with erythropoietin gene therapy approaches has demonstrated protection in models of Alzheimer’s and Parkinson’s disease. Now, we need to find a way to provide systemic delivery of erythropoietin microparticles so that they cross the blood-brain barrier or find a way to deliver them to the site of neurodegeneration through adeno-associated viruses.”