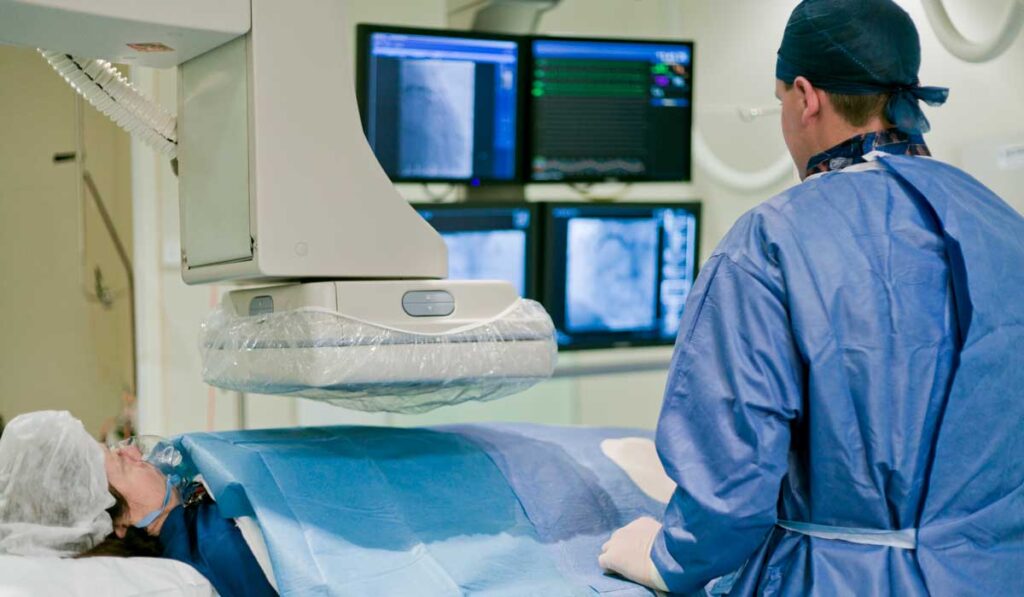
The versatile Shockwave Intravascular Lithotripsy (IVL) system has enjoyed expanding FDA approval for cardiovascular applications, beginning with peripheral artery disease (PAD) in 2016. It has since been used to treat calcific peripheral arteries and to facilitate transfemoral access for transcatheter aortic valve replacement (TAVR). Now, as of early 2021, it is approved to treat calcified coronary arteries.
Vanderbilt University Medical Center has been an early and rapid adopter of this technology. “Outcomes have been excellent and procedural time is cut in half, with patients often going home the same day,” said Colin Barker, M.D., director of interventional and structural cardiology at Vanderbilt.
Matthews Chacko, M.D., director of peripheral vascular interventions, has led the interventional team in applying IVL in PAD patients. “It is as effective as any sort of debulking, catheter-based atherectomy strategy that I’ve seen. It’s straightforward and safe, and it works over the same wire platform as any conventional intervention,” he said. “I think it has really revolutionized the management of calcific disease in both the coronary and the peripheral circulation.”
Restoring Diameter, Pliability
During IVL, the interventionalist positions the balloon in the calcified vessel stenosis. There it is inflated at a low pressure until it makes full contact with the vessel wall. The device then emits high-frequency ultrasound waves, which fragment the calcified plaque within the vessel wall.
“Once the calcific lesion has been fractured, the vessel enlarges and becomes much more pliable, which allows for safe passage of a stent – and now, even a valve,” Barker explained. “It replaces drilling inside arteries, which carries risks of vessel perforation or embolization that may cause small heart attacks. Also, it eliminates the need to provide mechanical circulatory support,” he said.
“I think it has really revolutionized the management of calcific disease in both the coronary and the peripheral circulation.”
The stakes are also lower because less pressure is needed to treat the calcification, lowering the risk of dissection and suboptimal stent expansion.
The improved pliability is particularly important with elderly patients. “When you are talking about putting a 14 French sheath that is about the size of a pinky finger into the calcified iliofemoral artery of an 80-year-old woman, the risks of vascular complications can be high. With IVL, we don’t have to resort to a sternotomy or higher risk alternative access,” Chacko said.
It can also limit the need for staged procedures in complex coronary patients. “If you remove the rotablator, it often requires taking the patient off the table and talking to them about the added risks of vessel perforation or dissection,” Chacko said. “IVL can be an ad hoc procedure. We aren’t adding much risk.”
Disrupt Trials Finishing Strong
Clinicians have not had to wait for a comprehensive mechanistic understanding of how these devices work. While numerous studies have explored the safety and efficacy of Intravascular Lithotripsy (IVL) in humans, the groundbreaking Disrupt trials have helped propel the technology over the finish line.
Findings from the 2020 Disrupt PAD III trial involving 306 patients with calcified PAD across 45 sites demonstrated a 15 percent higher procedural success rate for IVL versus standard percutaneous transluminal angioplasty, as well as lower arterial dissection rate and severity. They also saw a 75 percent relative risk reduction in the need for bail-out stenting, and a nearly four-fold lower stent implantation rate.
“With IVL, we don’t have to resort to a sternotomy or higher risk alternative access.”
The Disrupt CAD III trial enrolled 431 patients with calcified coronary artery disease. Findings following IVL included low rates of major periprocedural clinical and angiographic complications and a 92.4 percent procedural success rate.
Extravascular Applications
The benefits of IVL to facilitate transfemoral vascular access for TAVR are well described. Now, applications outside the vessel walls are on the horizon. “The mitral valve is prone to calcium buildup, which can be very hard to deal with during a catheter-based procedure,” Barker said. “IVL may provide a way to modify the calcium in and around the valve. We’re really excited about how that might improve leaflet function.”