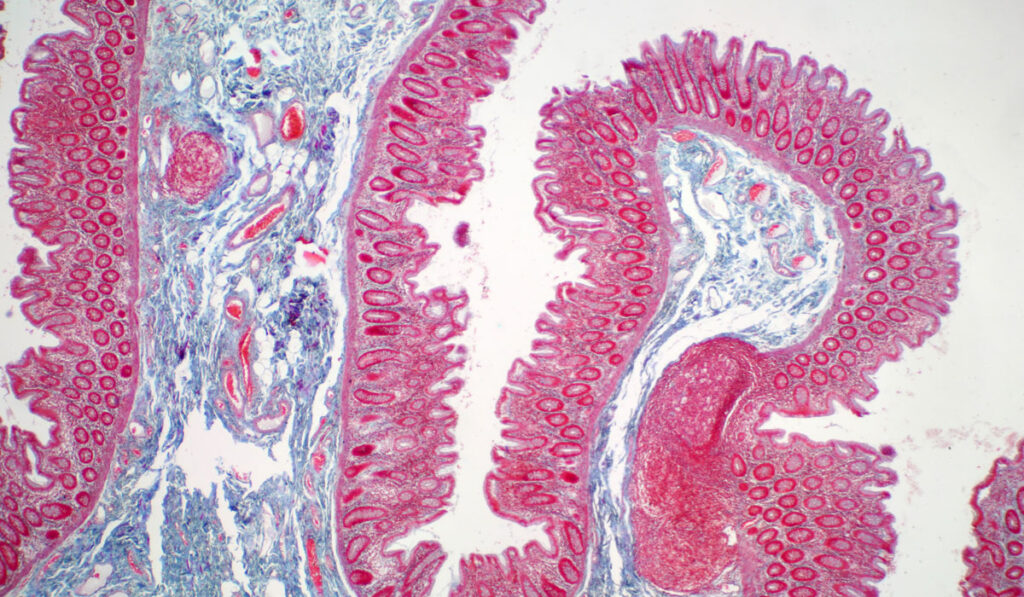
Altered barrier function is thought to play a role in celiac disease (CeD), but there has been no reliable approach to measuring this function in duodenal mucosa. That may change with a novel, catheter-based mucosal impedance (MI) technology originally developed for esophageal applications by gastroenterologist Michael Vaezi, M.D., of Vanderbilt University Medical Center.
In a study published in Clinical Gastroenterology and Hepatology, specialists at Vanderbilt’s Digestive Disease Center evaluated the feasibility, safety and clinical utility of the new technology to measure duodenal impedance in patients with CeD.
“We have needed better diagnostic tools for celiac disease,” said Dawn Adams, M.D., director of Vanderbilt’s Celiac Disease Clinic. “Currently, we have to biopsy the small intestine to definitively diagnose adults. It is invasive and they must be consuming gluten before the test. The MI catheter may allow us to diagnose earlier and find mucosal changes that are not seen on biopsy.”
“People with active celiac disease appear to have different mucosal impedance values than those with inactive disease.”
MI for Celiac Diagnosis
The direct MI approach is based on identifying a unique signature of change in electrical signals. Using a novel probe placed through the working channel of an endoscope, mucosal conductivity is measured directly through surface contact.
The study enrolled patients with CeD and controls undergoing esophagogastroduodenoscopy (EGD) for other indications. All patients underwent EGD with MI and biopsy. Four MI measurements were obtained in the second portion of the duodenum and two in the bulb (6 and 12 o’clock) followed by corresponding biopsies. Biopsies were interpreted by gastro-intestinal pathologists blinded to the MI results and reported as Marsh scores.
The results confirmed Adams’ hypothesis: “People with active celiac disease appear to have different mucosal impedance values than those with inactive disease,” she said.
Benefits of MI Measurements
The study showed the feasibility of employing the MI device during endoscopy as a potential marker of disease activity. The catheter added almost no risk to a standard endoscopy and little extra time, the researchers found.
The MI catheter can provide a real-time measurement during endoscopy; there is no added cost for biopsy interpretation, Adams said. The objective numerical output has less potential for reader variability and offers the possibility of severity grading for disease activity. Additionally, the MI probe can quickly take several measurements, which may improve diagnosis.
“Higher MI in the inactive CeD group suggests that mucosal abnormalities exist that are missed with biopsy and could play a role in diagnosing potential CeD or difficult-to-diagnose populations,” Adams added.
Setting the Stage for Future Research
The Vanderbilt Celiac Disease Clinic has been recognized by the Society for the Study of Celiac Disease (SSCD) for its combination of patient care and dedication to research. The clinic is one of a handful of major centers in the U.S. to offer a continuous care model for the treatment of CeD and refractory CeD.
Adams is establishing a tissue repository from CeD patients to prospectively collect samples for future research. Through patient surveys, she has begun collecting data on quality of life, CeD-related stress, and diet compliance.
“Higher MI in the inactive CeD group suggests that mucosal abnormalities exist that are missed with biopsy.”
Using additional patient survey data, researchers at the center are investigating the prevalence of ARFID among patients with CeD. ARFID is a newly identified eating disorder in which a patient’s eating restrictions are not due to fear of body weight or shape, “but rather by excessively picky eating, limited appetite or lack of interest in eating, and fear of negative consequences, such as choking, vomiting, or gastrointestinal distress from eating,” Adams said. She suspects this is highly prevalent in patients with CeD who have suffered from GI symptoms with eating gluten prior to diagnosis.
The clinic is also participating in two multicenter phase 3 clinical trials for new drugs to treat celiac disease. One trial is evaluating larazotide acetate for the relief of CeD symptoms and the other, PRV-015 in adult patients with non-responsive celiac disease who are on a gluten-free diet.
“Celiac disease affects about 1 percent of our population and the public is becoming more aware of its many manifestations,” Adams said. “We’re in a unique position because our patients can inform our research and our research can translate directly to the clinic.”