While jaundice is typically benign, if untreated it can lead to encephalopathy, kernicterus and potentially irreversible brain damage. Monitoring of jaundiced neonates requires measurement of total serum bilirubin in blood samples. Transcutaneous bilirubinometry (TcB) machines measure skin yellowness and are typically used as screening tools to limit painful blood draws.
However, in areas with limited resources, including Sub-Saharan Africa and South Asia, commercially available TcB devices can be cost prohibitive. Newborns in remote locations or in families with cultural norms that preclude hospital visits often go without this screening.
Under a Bill and Melinda Gates Foundation grant, collaborators Jörn-Hendrik Weitkamp, M.D., an associate professor of pediatrics and director of Patient-Oriented Research at Monroe Carell Jr. Children’s Hospital at Vanderbilt, and Chetan Patil, Ph.D., an assistant professor of bioengineering at Temple University, have developed a smartphone-based “phoneTcB” that targets this at-risk population.
“Screening of these jaundice in newborns is vital, since timely access to effective phototherapy is key to curbing bilirubin-induced mortality and morbidity in high-burden countries,” Weitkamp said. “The ultimate purpose of phoneTcB is to improve neonatal health worldwide by making it accessible to anyone with a smartphone camera.”
“Screening of these newborns is vital, since timely access to effective phototherapy is key to curbing bilirubin-induced mortality and morbidity in high-burden countries.”
Risks of Undetected Jaundice
Internationally, pathological neonatal jaundice accounts for 1,309 deaths per 100 000, and ranks seventh among causes of mortality in the first week after birth. The risk of death is highest between 3 and 6 postnatal days, when plasma or serum bilirubin levels typically peak.
Since bilirubin is excreted through the urine and stool, Weitkamp says hot climate and poor maternal nutrition in some regions can escalate the severity and persistence of bilirubinemia. The genetics of ethnicity and race may also contribute to more severe complications from jaundice in newborns due to higher incidence of G6PD deficiency and sickle cell disease among African Americans, for example.
A Low-cost, Portable Screening Device
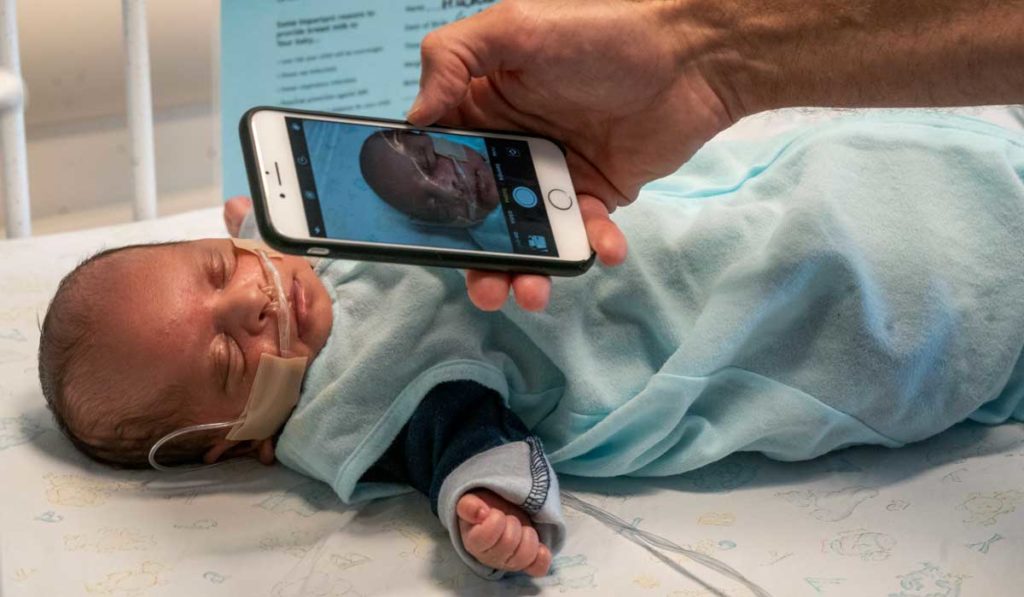
Weitkamp says phoneTcB operates on similar principles to TcB machines routinely used by hospital nurseries for jaundice screening, but at a fraction of the cost and with optics that can be added to typical smartphones used around the world.
To collect accurate measurements, the system uses filters that only allow light at particular wavelengths to pass. The camera lens and flash are brought into direct contact with the skin and measurements are acquired from the forehead or sternum in a few seconds. The flash output of the phone is calibrated by taking serial pictures of reference grey, white and black cards. Measurements are sent via app to offline custom software that calculates the bilirubin count.
Testing the Technology
Weitkamp’s team has tested phoneTcB screenings with about 50 neonates at Children’s Hospital within two hours of blood draws. The data show high correlation between blood bilirubin levels and smartphone predictions. This was consistent across the major demographics in the group.
Additional studies are now underway at collaborating sites in Tanzania and Nigeria investigating the influence of different skin pigmentation and calibration methods. To date, data on full-term infants of African origin across all sites are similar to findings obtained from Caucasian infants, showing high correlations with blood bilirubin levels.
Still to be assessed is how accurate the phoneTcB is across the wider, more variable range of infant skin conditions that exist in older or premature babies.
Deployment Challenges
Future work could lower the device’s cost and make it more accessible, Weitkamp says. The phoneTcB system could be redesigned to eliminate the need for the accessory filters. Moreover, there is a question of how to connect infants to treatment in low-resource settings if they receive an abnormal screening result.
Weitkamp envisions expanding the smartphone-based system to other clinical screenings. “There is a big need for tools that allow for non-invasive measurements on our most vulnerable population,” Weitkamp said. “Devices such as the phoneTcB can be developed to monitor other problems commonly affecting preterm babies, such as anemia or heart rate changes.”