Research from Jörn-Hendrik Weitkamp, M.D., an associate professor of pediatrics at Monroe Carell Jr. Children’s Hospital at Vanderbilt, is shedding light on the role of a specific type of immune cell – regulatory T cells – in necrotizing enterocolitis (NEC) pathology.
“Regulatory T cells seem to be very important to maintain a successful pregnancy, and for the fetus to build a tolerance to paternal antigens in utero,” Weitkamp said.
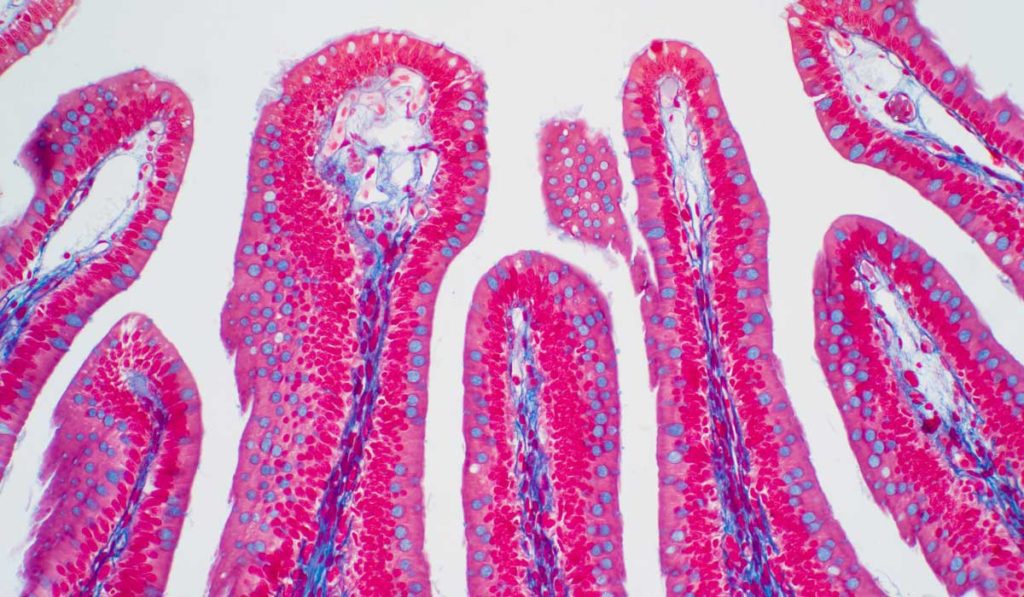
NEC, the most common surgical emergency in preterm infants, is thought to occur when pathogenic bacteria invade an immature intestinal barrier, eliciting inflammation that spurs disease development. While the underlying cause is unclear, numerous studies point to inappropriate immune responses as playing a central role.
“Regulatory T cells seem to be very important to maintain a successful pregnancy, and for the fetus to build tolerance to paternal antigens in utero.”
Revealing the Importance of Regulatory T Cells
Despite their importance in many human diseases and prevalence in the intestinal mucosa, T cells’ role in the pathogenesis of NEC was overlooked for a long time due to differences between the animal models used to study the disease and humans.
Said Weitkamp, “We discovered early that the ontogeny and development of the cellular immune system in the neonatal mouse is very different from a neonatal human.”
Weitkamp has shown that regulatory T cells in human infants are abundant at an early gestational age. In contrast, “most models for NEC are in neonatal mice. And neonatal mice barely have any of these cells present when they’re born,” Weitkamp said.
T Cell Ratios in NEC
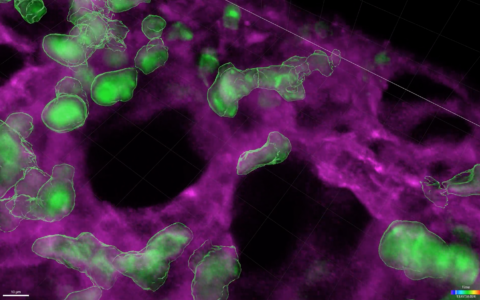
To study whether regulatory T cells play a role in NEC pathology, Weitkamp collaborated with surgeons and pathologists at Vanderbilt to analyze T cell populations from infants with and without NEC.
Their findings, published in the journal Gut, revealed that the relative abundance of regulatory T cells in the intestines of infants with NEC is significantly lower than in infants without NEC. Their studies also revealed that between initial resection for NEC and reanastomosis, levels of regulatory T cells can recover.
Related research from the group published in PLoS ONE and Immunity indicates that another type of T cell, intraepithelial lymphocytes (IELs), which can be found wedged between cells of the intestinal epithelium, are also selectively reduced in infants with NEC.
Collectively, the findings suggest that depletion of specific types of T cells contributes to the inflammatory sequelae associated with NEC, and that promoting the development of these cells may be a potential therapeutic strategy.
Ongoing Research
The next pressing topic for Weitkamp and his research team is to understand what drives the deregulated immune response in NEC. Their ongoing research is revealing that two known regulators of T cell function – SMAD7 and TGF-ß – may provide a mechanistic link.
“We have some evidence that there may be some maternal immune activation that increases SMAD7 to cause a decrease in T regulatory cell induction and the decrease of IELs in the preterm intestine, which then makes them more vulnerable to develop the disease if there’s another trigger,” Weitkamp said.
The team is currently completing studies to confirm these findings for publication.