Atezolizumab, a checkpoint inhibitor that reverses T cell inhibition by blocking PD-L1, was FDA-approved last year for the treatment of triple-negative breast cancer (TNBC). This extended to only those TNBC patients whose tumors expressed PD-L1, which comprised 40 percent of the population enrolled in the study leading to approval.
A multicenter study led by Vandana Abramson, M.D., associate professor of medicine in the Division of Hematology and Oncology at Vanderbilt University Medical Center, seeks to better identify which patients could potentially benefit. The trial investigates atezolizumab in combination with carboplatin, chemotherapy used for TNBC.
“We are trying to determine mechanisms of both response and resistance to atezolizumab,” Abramson said. “This is so important because, to date, TNBC patients have few good treatment options and none that are not chemotherapy-based or provide a durable response. Biomarkers of response to a drug like atezolizumab need to be better understood.”
“TNBC patients have few good treatment options, and none that are not chemotherapy-based or provide a durable response. Biomarkers of response to a drug like atezolizumab need to be better understood.”
TNBC Subtyping to Identify Responders
Abramson says that certain TNBCs are “immune cold” due to the absence of immune cells in the tumor and enrichment of immune suppressive cells in the microenvironment. Chemotherapy such as carboplatin works to change this by disrupting DNA in cancer cells, triggering the creation of neoantigens. Checkpoint inhibitors help on a second front by modulating the tumor immune response.
Understanding which TNBC subtypes respond to checkpoint inhibitors is crucial in determining which drug combinations are likely to work best in which subsets of patients. The study will rely on the work of Benjamin F. Byrd, III, professor of medicine, professor of oncology and Vanderbilt-Ingram Cancer Center director Jennifer Pietenpol, Ph.D., and Brian Lehmann, Ph.D., research assistant professor of medicine at Vanderbilt, who classified TNBC into four subtypes: basal-like (BL1 and BL2), mesenchymal, and luminal androgen receptor. Researchers will gauge responses to therapy by these subtypes.
Learning from Biopsies
Study participants will be randomized to receive either atezolizumab with carboplatin or carboplatin alone. Participants whose disease progresses on carboplatin alone can cross over to atezolizumab.
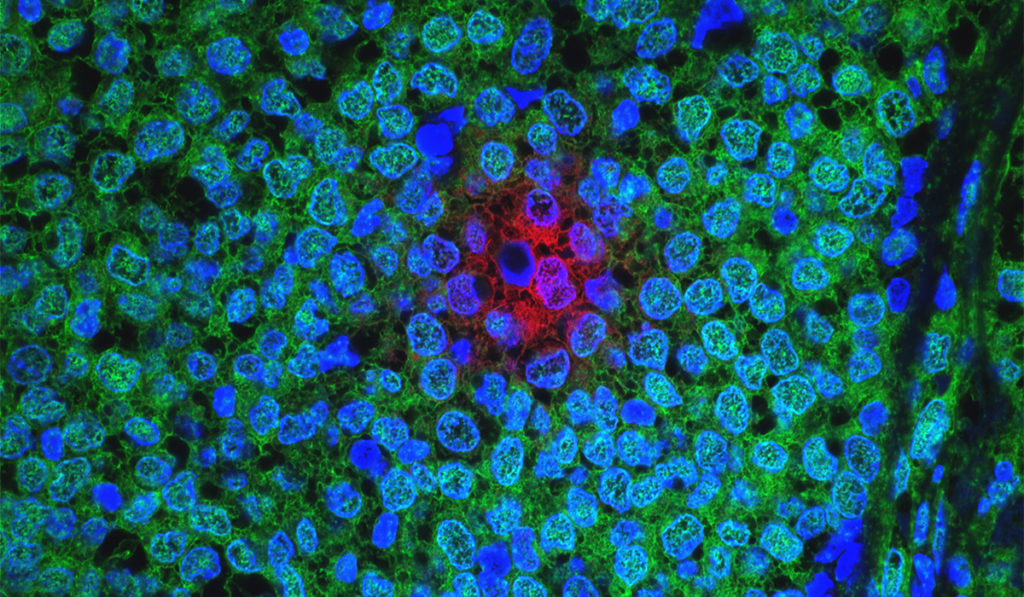
A biopsy of a metastatic site will be performed at baseline and upon disease progression. These biopsies will enable the team to evaluate biomarkers such as tumor-infiltrating lymphocytes, TNBC subtype, BRCA 1/2 status, immune cell infiltrate percentage, and PD-L/PD-L1 status.
“The biopsies are a central part of this study because tissue analysis will give us information regarding why certain patients are responding to the treatments and others are not, ultimately helping us develop biomarkers of response and resistance,” Abramson said.
“Tissue analysis will give us information regarding why certain patients are responding to the treatments and others are not.”
A Goal of Personalized Therapy
Atezolizumab works by inhibiting PD-L1 on T cells and thereby upregulating immune response. The New England Journal of Medicine study leading to FDA approval looked at atezolizumab plus nab-paclitaxel in TNBC patients and found improvements in progression-free survival and overall survival in the PD-L1 positive subset. Abramson is including not only the approximately 60 percent of TNBC patients who don’t express PD-L1, but also those expressing PD-L1.
“We hope this research will refine biomarkers of response so that those who are likely to benefit will be able to receive this treatment and those who are unlikely to benefit will be spared the potential toxicities,” Abramson said.
The study has enrolled 90 patients at eight academic hospitals around the country so far, Abramson said, but recruitment is paused until the end of restrictions created by COVID-19.