In a new meta-analysis of observational studies and randomized controlled trials (RCT), a team led by Vanderbilt University Medical Center researchers provides a comprehensive summary of existing data on the cardiovascular adverse effects of traditional androgen deprivation therapy (ADT).
Following an initial analysis in 2006 of the Surveillance, Epidemiology, and End Results (SEER) Medicare database, there have been growing reports linking ADT for prostate cancer and adverse effects on the cardiovascular system. These reports led to a science advisory in 2010 on the increased cardiovascular risks of ADT.
The latest analysis is “a comprehensive review of all the ADTs involved in the care of prostate cancer,” said Joshua Beckman, M.D., co-author and director of vascular medicine at Vanderbilt. Adding, “We assessed the existing data in the literature and closely examined some of the discrepancies, especially among patients in trials, who tend to be healthier, compared to ‘real world’ populations, where patients have cardiovascular comorbidities.” Javid Moslehi, M.D., is a senior author on the study and director of the cardio-oncology program at Vanderbilt.
Associations from Observational Studies
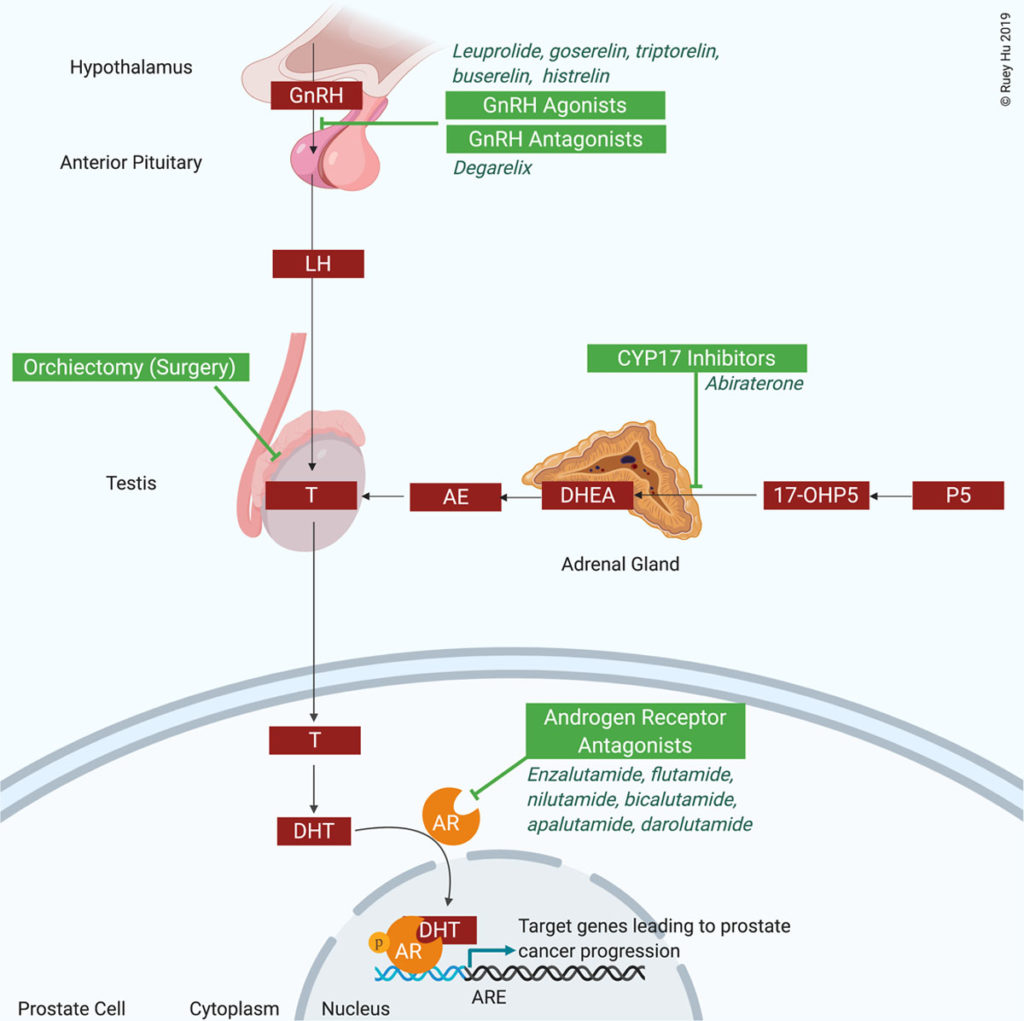
The analysis found that ADTs had positive associations with cardiovascular events, cardiovascular death and myocardial infarction among three meta-analyses of observational studies, but not among three meta-analyses of RCTs.
“This discrepancy corroborates something that has been suspected by clinicians for a number of years, and illustrates why it is so important for meta-analyses and large-scale trials to be conducted on this topic,” said first author Jiun-Ruey Hu, M.D., an internal medicine resident at Vanderbilt.
“This discrepancy corroborates something that has been suspected by clinicians for a number of years.”
Different Analysis Strategies
RCTs may not capture an association of ADT with cardiovascular adverse events for several reasons. These include the fact that RCT primary endpoints are measures of prostate cancer control and not cardiovascular events, and that standard practice for enrollment is to exclude patients with high risk, including high cardiovascular risk.
When assessed as individual ADT classes, gonadotropin-releasing hormone (GnRH) agonists, GnRH antagonists and combined androgen blockage had either mixed or positive associations with cardiovascular adverse effects among meta-analyses of observational studies. Notably, abiraterone, a CYP17 (cytochrome P450 17A1) inhibitor, stood out as having positive associations with cardiovascular events and hypertension in two meta-analyses of RCTs.
Understanding the Risks
In addition to serving as a resource for physicians, Beckman says the new analysis reflects two topics of broad interest to the medical field.
“For one, we are trying to pick out a population of people who won’t benefit from standard therapies, and that is a topic which applies for other drugs and diseases as well. In addition, this review highlights mechanisms by which common adverse problems, like insulin resistance, diabetes or cardiovascular disease, develop through separate pathways, which helps us understand the physiology better.”
Added Hu, “It’s important for clinicians and patients to be able to access quantitative data about side effects of the treatments so they can make informed decisions about their cancer therapies.”