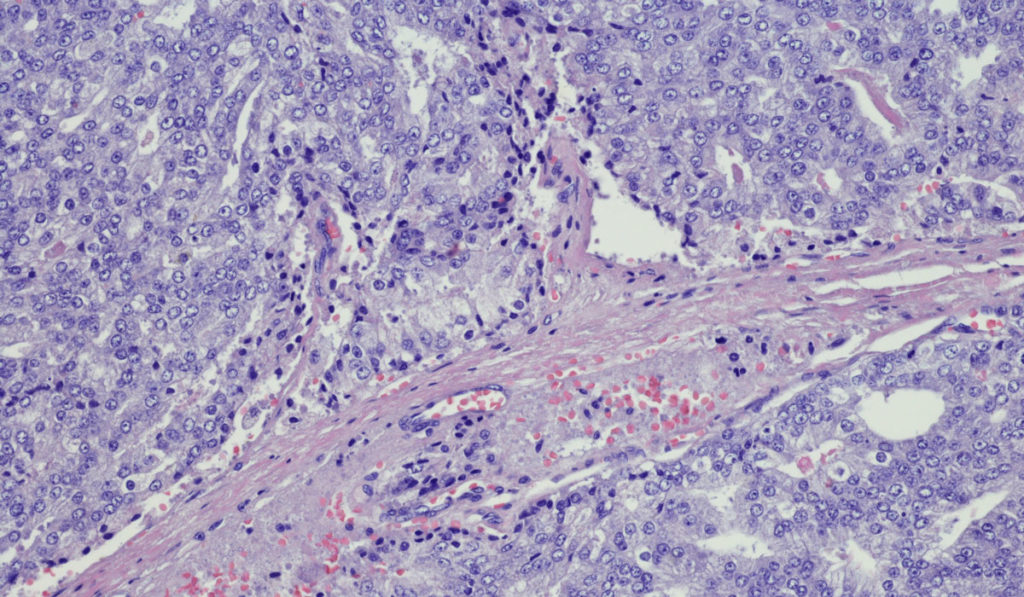
Advanced prostate cancer treatment has seen an explosion of novel agents in the last 15 years, starting with chemotherapeutics and expanding further with the arrival of androgen receptor antagonists and checkpoint inhibitors.
At one time, androgen deprivation therapy (ADT) was the only option for treating metastatic prostate cancer. Using surgery or drugs such as leuprolide or goserelin, ADT halts the body’s production of testosterone.
“That was the foundation of treatment for years,” said David Penson, M.D., professor and chair of the Department of Urology at Vanderbilt University Medical Center. “If a patient had metastatic prostate cancer, we would give him ADT and maybe radiation to where the cancer spread. The ADT worked for a few years, but eventually, it would stop working and the cancer would spread.”
Chemotherapy and Immunotherapy
The expansion of prostate cancer treatment options started with traditional chemotherapy. “First you had mitoxantrone, which didn’t prolong life, but it did help with symptoms in men with disease which no longer responded to standard hormonal therapy,” Penson said. “Then came docetaxel, which prolonged survival by about three to four months – not insignificant for someone who had a two-year average life expectancy at the time.”
After that, the immunotherapy drug sipuleucel-T hit the market. “It also prolonged life by about four months,” Penson said.
“The model has always been, if you have activity with aggressive disease, you’ll likely have activity at earlier stages and with less aggressive tumors.”
Androgen Receptors and Checkpoint Inhibitors
Advances in the last five years have centered around the androgen receptor antagonists enzalutamide, apalutamide and darolutamide, and the hormone therapy drug, abiraterone. These new agents have been shown to improve outcomes for castration-resistant prostate cancer, and now the focus is on determining whether the drugs improve outcomes for the hormone-sensitive disease.
The ENZAMET and ARCHES trials studying enzalutamide, and the TITAN trial studying apalutamide, have shown that the addition of either agent to ADT improves outcomes in men with metastatic hormone-sensitive prostate cancer. “A clinical medical castration removes 95 to 97 percent of the testosterone,” Penson said. “You then add these additional agents, and you get rid of the rest.”
“The model has always been, if you have activity with aggressive disease, you’ll likely have activity at earlier stages and with less aggressive tumors,” Penson said.
A similar trend is being observed with checkpoint inhibitors, like pembrolizumab, said Penson. “We’re using them in the worst stages and the worst disease and we’re starting to see a response; then we move them upstream.”
Penson says future studies need to focus on dosing and sequencing to fine-tune use of these novel agents.
Testing a New Concept
In a new direction for prostate cancer care, ongoing studies like the TRoMbone trial are assessing whether there is a benefit to treating minimal metastatic disease both systemically and locally, using radiation or surgery.
“That’s a complete paradigm shift in the way we treat prostate cancer. It’s certainly not the standard of care at this point, but it’s a new concept being tested in trials.”
“For a patient where the cancer has spread, for example to their back or lymph nodes, the thought was there is no benefit to treating the primary tumor,” Penson explained. “Now, what we’re finding is that for men who have minimal spread elsewhere in their body – one to three lesions – when we use these novel agents and treat the primary tumor at the same time, they do even better.”
“That’s a complete paradigm shift in the way we treat prostate cancer. It’s certainly not the standard of care at this point, but it’s a new concept being tested in trials.”