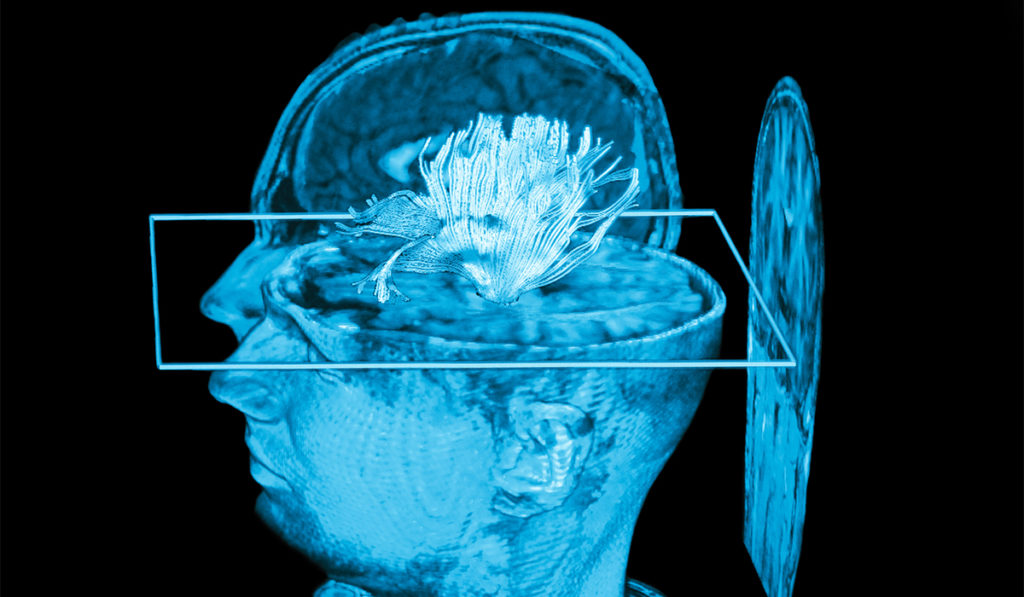
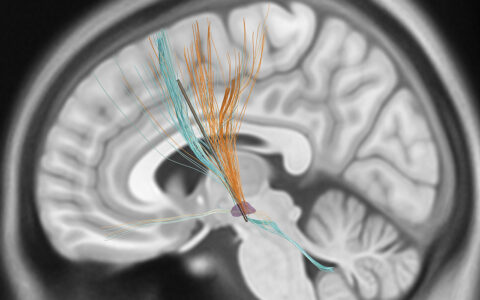
Focused ultrasound (FUS) is finding increasing applications in the brain and body due to its unique ability to non-invasively activate and inhibit neurons. While this has raised hope of a non-opioid, non-addictive treatment for chronic pain, no studies have looked at FUS for pain management. To translate its use to humans, researchers are developing technologies to guide treatment and dosing.
With funding from the National Institutes of Health, awarded as part of the Helping to End Addiction Long-term Initiative, a team of researchers from the Vanderbilt University Institute of Imaging Science has established pre-clinical tools to use FUS to modulate and functionally monitor brain circuits, and are scaling this technology to humans with the goal of modulating brain circuits that underlie pain perception.
“Ultrasound neuromodulation is an exciting area because it allows you to alter activity non-invasively, with fine spatial precision, in deep or superficial brain targets.”
“While other devices to treat pain exist, their efficacy is limited by inaccurate targeting of pain regions and circuits in the brain,” said William Grissom, Ph.D., associate professor of biomedical engineering at Vanderbilt and a principal investigator on the project. “Ultrasound neuromodulation is an exciting area because it allows you to alter activity non-invasively, with fine spatial precision, in deep or superficial brain targets.”
Targeting Specific Pain Nodes
Deep brain stimulation and ablation procedures are currently used clinically, targeting the thalamic nuclei, ACC, and PAG/PVG regions of the cortex, but there are technological challenges. The researchers hope to use FUS neuromodulation to accurately and reliably stimulate these brain regions associated with pain.
“With device-based solutions, it is challenging to know prior to implantation whether the pain node being stimulated will alleviate chronic pain symptoms,” said Charles Caskey, Ph.D., lead principal investigator on the project and assistant professor of biomedical engineering, radiology and radiological sciences at Vanderbilt. “We hope that understanding how different neuromodulatory effects at various targets affect pain will improve the efficacy of existing methods and guide the development of improved treatments.”
Device Design
The researchers previously developed and laboratory tested an MRI-guided focused ultrasound system for modulating neural activity in the somatosensory cortex. They now plan to work alongside Peter Konrad, M.D., professor of neurological surgery at Vanderbilt, to further develop the device for clinical use. Other Vanderbilt collaborators include Li Min Chen, M.D., assistant professor of radiology and radiological sciences and a co-PI on the project, John Gore, Ph.D., Hertha Ramsey Cress Chair in Medicine and director of the Vanderbilt University Institute of Imaging Science, and Benoit Dawant, Ph.D. director of the Vanderbilt Institute for Surgery and Engineering.
The device will resemble an MRI head coil and combine functional MRI with FUS neuromodulation. It will allow the researchers to navigate to precise brain regions under MRI guidance and to monitor the responses of pain circuits using a functional MRI (fMRI) readout.
Grissom hopes that the device will become a viable alternative to long-term opioid use for chronic pain. “Device-based treatments offer an alternative that may eliminate risks such as addiction and drug diversion,” he said. “By the end of the project, we expect to have a fully developed and validated an MRI-guided ultrasound system that is ready for pilot clinical trials in various pain management applications.”