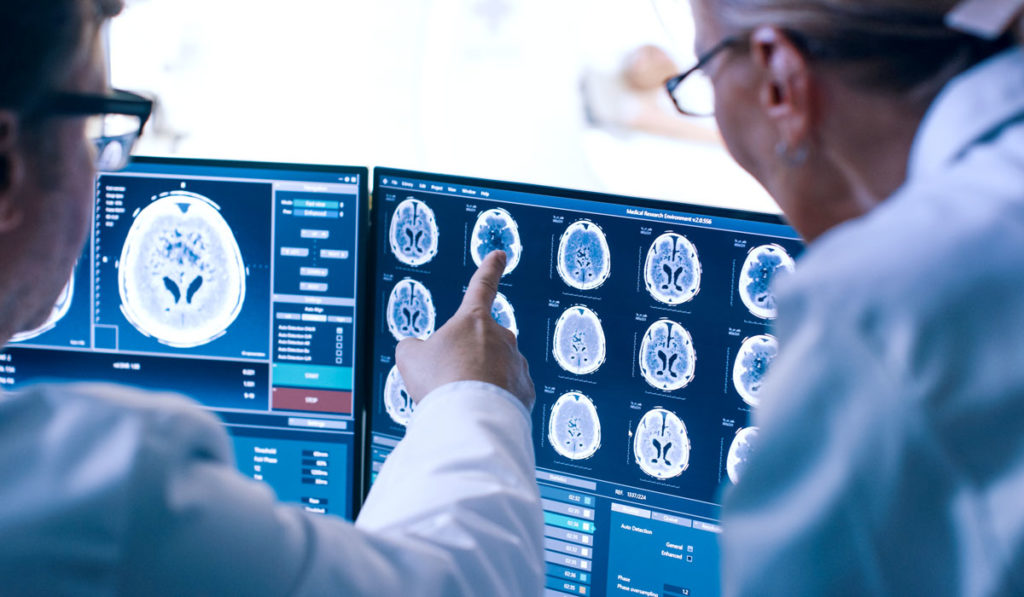
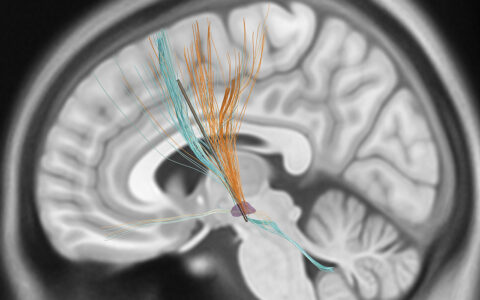
Measuring the volume of the dentate nucleus is important for tracking the progression of Parkinson’s disease and essential tremor, and may be useful in mapping implant surgery. However, it is challenging to elucidate deep gray matter via MRI, so volumetry assessment is currently a time-intensive process that must be tailored to each patient’s brain.
Neurologist Daniel Claassen, M.D., of Vanderbilt University Medical Center and Camilio Bermudez, an M.D./Ph.D. candidate at Vanderbilt, are employing machine learning to develop an algorithm that performs these delineations in short order. They have already demonstrated that the algorithm can replicate the accuracy and reliability of manual segmentation.
“The system we developed can delineate the dentate nucleus in such a way that when you present it with a new image of a new person, it can find it without any other hints,” Claassen said. He and Bermudez presented their research at the American Neurology Association Annual Meeting in October 2019.
Targeting the Imaging Deficit
None of the three types of MRI (T1-weighted, T2-weighted and diffusion-weighted) are effective at delineating the dentate nucleus, Claassen explained, so determining volume and the anatomical location is currently an exercise in elimination. Neurologists must identify surrounding landmarks, such as the cerebrospinal fluid, white matter tracks, and deep cerebellar nuclei, essentially looking for where the dentate nucleus is not. Since every brain is different, this can be a protracted process.
“The system we developed can delineate the dentate nucleus in such a way that when you present it with a new image of a new person, it can find it without any other hints.”
“As technology is evolving, we realize there are all kinds of problems with aligning two brains perfectly,” Bermudez said. Their approach addresses this by machine learning intensity patterns from a large sample of brain MRIs. With enough iterations, the dentate nucleus and surrounding structures can be readily identified.
Claassen and Bermudez first created a large data set of 100 cerebellar images where the dentate was manually identified using multimodal MRI methods. They then trained a deep neural network algorithm to recognize the patterns made by the brain structures and become “smart” about delineating these regions. “Now, instead of having a human go in and delineate these structures across all modalities, we can perform it automatically in a matter of seconds,” Claassen said.
Future Applications
The algorithm is not only useful in tracking disease progression. It may also be used to localize structures for applications like deep brain stimulation implants. “Some clinical cases suggest that targeting the dentate nucleus for a stimulation device helps with spasticity following stroke. Using this technology could help in pre-surgery planning to optimize placement of the device,” Bermudez said.
The approach holds a broader promise for enhancing clinical acumen. Already machine learning has helped oncologists identify cancerous skin lesions from patient photos.
Other neurology applications may include studying changes in the hippocampus in Alzheimer’s and epilepsy patients. “This is an area that is very ripe for discovery and translational research, bringing these deep learning and image processing ideas and tools that exist in the research realm to assist patient care,” Claassen said.