The “blue” half of the cardiovascular system – the venous system – has traditionally received less attention and research investment than its “red” counterpart. Yet venous thromboembolism (VTE) remains the third most common cardiovascular diagnosis, with $200 million dollars spent treating post-thrombotic syndrome in the U.S each year. In 2008, the U.S. Surgeon General issued a Call to Action to promote VTE prevention. From that charge, the NIH began funding enhanced research on the disease.
Preclinical models are of critical importance in this research, but standardization has been lacking, making it difficult to compare results across laboratories. Under the leadership of José Diaz, M.D., director of the Division of Surgical Research at Vanderbilt University Medical Center, the American Venous Forum recently issued a new consensus assessment that offers clarity for researchers using preclinical models.
“I saw a huge need to clarify the surgical techniques we were using to generate venous thrombus (VT), to provide guidance for researchers on choosing an appropriate model for their research – a commonly confounding issue,” Diaz said.
Diaz spoke on “Frontiers in Thrombosis: Progress in Venous Thromboembolism” at the American Heart Association Scientific Sessions on November 18, 2019.
From Presumption to Certainty
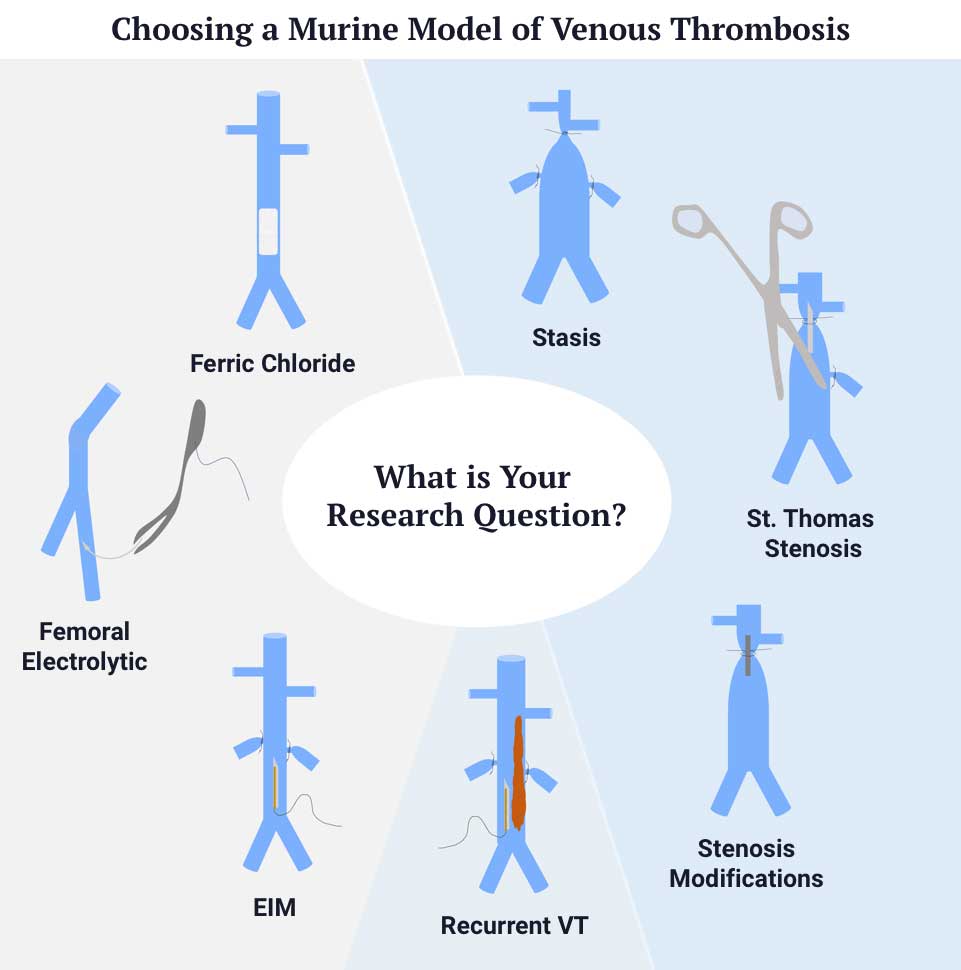
Diaz assembled a task force composed of ten leaders in the field to describe the surgical technique, advantages, and disadvantages of seven common models. They developed an algorithm to aid in choosing a model based on the research question. “This provides clear decision matrix for researchers and grant reviewers,” Diaz said.
“This provides clear decision matrix for researchers and grant reviewers.”
The new consensus includes detailed definitions of materials and methods that describe stasis (ligation), St. Thomas stenosis, stenosis modifications, electrolytic inferior vena cava (IVC), ferric chloride, femoral vein electrolytic and recurrent models. The surgical technique section details important factors such as the interruption of branches draining into the IVC, the choice of spacer in the stenosis models, and application of current in electrolytic models. “These VT models were chosen based on their use and thorough characterization to enable reproducibility between laboratories,” Diaz said.
Describing and delineating these models enables technique comparisons. “Before, you might find two studies on the same drug using the ‘stasis’ model, one using a certain ligation method that may result in a 12 mg blood clot, another using a method that results in a 25 mg blood clot. You could find the drug works in the one case but not the other, but you would be comparing apples and oranges.”
A Novel Research Tool
Endorsed by the International Society on Thrombosis and Haemostasis and the American Heart Association, the consensus article is the first of its kind in the VT field. The utility of the consensus extends beyond establishing a common language among researchers. Grant reviewers and panel members at the NIH can use it in evaluating proposals and studies.
“Reviewers will have a tool they can download and use in their questioning,” Diaz said. “You don’t want to assign a million-dollar grant and find out the investigator has the wrong model for what they are trying to do.”
The bigger picture for Diaz is the need to support basic and applied research for VT. “There is a need to better understand the mechanisms involved in VT. Preclinical models are key to understanding this, but the models themselves must be well-characterized and consistent across laboratories for true advancements to be made. Reproducibility is key, and this consensus gives us the tools to make our research comparable, reproducible, and ultimately more effective.”
“The models themselves must be well-characterized and consistent across laboratories for true advancements to be made.”