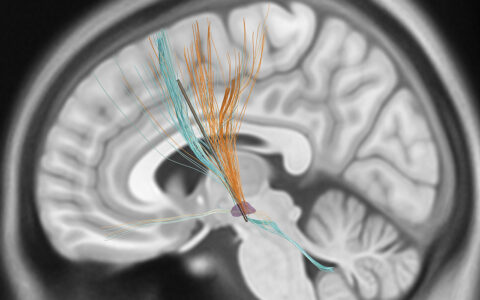
Animal studies have suggested that a glymphatic system exists in the brain similar to the lymphatic system in the body. This recently discovered system may function as a waste-clearance pathway involving fluid communication between paravascular spaces, interstitial fluid and meningeal lymphatic vessels. It may be dysfunctional in patients with age-related neurodegenerative disorders such as Parkinson’s disease (PD) and Alzheimer’s, possibly related to an accumulation of cerebral amyloid.
A new study at Vanderbilt University Medical Center, funded by the Department of Defense, will combine the glymphatic system hypothesis with a growing body of research showing that PD patients benefit neurologically from exercise. The study will use novel neuroimaging methods to evaluate the glymphatic function and beta-amyloid burden response in PD patients participating in a 12-week, community-based exercise course.
“Early research indicates glymphatic functioning improves following exercise.”
“Early research indicates glymphatic functioning improves following exercise,” said Daniel Claassen, M.D., associate professor of neurology at Vanderbilt University Medical Center and principal investigator on the study. “We thought it would be an interesting intersection to understand how involvement in a community exercise class could potentially regulate or improve glymphatic flow in the brain and mitigate neurobehavioral response.”
Boxing as PD Therapy
Claassen noticed that among patients of the Vanderbilt Movement Disorders Clinic, there was an uptick of interest in the “Rock Steady Boxing” exercise class. “A number of our patients attended it,” he said. “Many spoke highly of the benefits they experienced from the class. Some described better gait, better mood or more clarity of thought. We wanted to explore that further.”
The study will assess patients with a clinical diagnosis of PD during, prior and following their participation in a 12-week Rock Steady program, assessing neuroimaging (beta-amyloid deposition, glymphatic flow), and clinical severity (motor, cognitive, sleep disturbance, mood). The primary outcome will be an MRI-based measurement estimating glymphatic diffusion.
Imaging Glymphatic Flow
Currently, the connection of the glymphatic system to PD or other neurodegenerative diseases is not well understood. “Previously, it had been thought that fluid in and around the brain operates independently. This has opened up a whole new way of thinking about the natural history of neurodegenerative disease, particularly as it pertains to waste clearance, which appears to be the main function of glymphatic flow,” said Colin McKnight, M.D., assistant professor of radiology and radiological sciences at Vanderbilt.
Relevant to the boxing study, McKnight and colleagues have been exploring imaging tools that can quantify cerebral blood flow, cerebrovascular reactivity (CVR) and CVR compliance time (the time required for arterioles to respond maximally to vasoactive stimuli) in a clinical setting. The boxing study will also use diffusion tensor imaging to observe slow diffusion of water molecules in the brain.
“We’ll be assessing glymphatic flow using noninvasive technologies,” he said. “As an exploratory method, we’ll look at glymphatic drainage in the cervical vessels. Then we’ll look higher up to see if we can follow glymphatic flow in the brain.”
Moving Toward Understanding
“This may have important consequences for how we understand aging and neurodegeneration.”
Claassen and McKnight are collaborating with colleagues across Vanderbilt, from nuclear medicine to physical rehabilitation to patient engagement. “It takes so many kinds of expertise to put this type of research project together,” Claassen said.
He added, “If we’re able to show that exercise does improve the clearance of pathologic proteins in the brain, it could be a really profound step toward understanding the different mechanisms of neurodegeneration and mechanisms of therapies, which have been largely difficult to prove in the current environment.”
“If our hypothesis is correct,” McKnight said, “this may have important consequences for how we understand aging and neurodegeneration. This may shed light on why particularly problematic proteins are cleared poorly from the brain.”