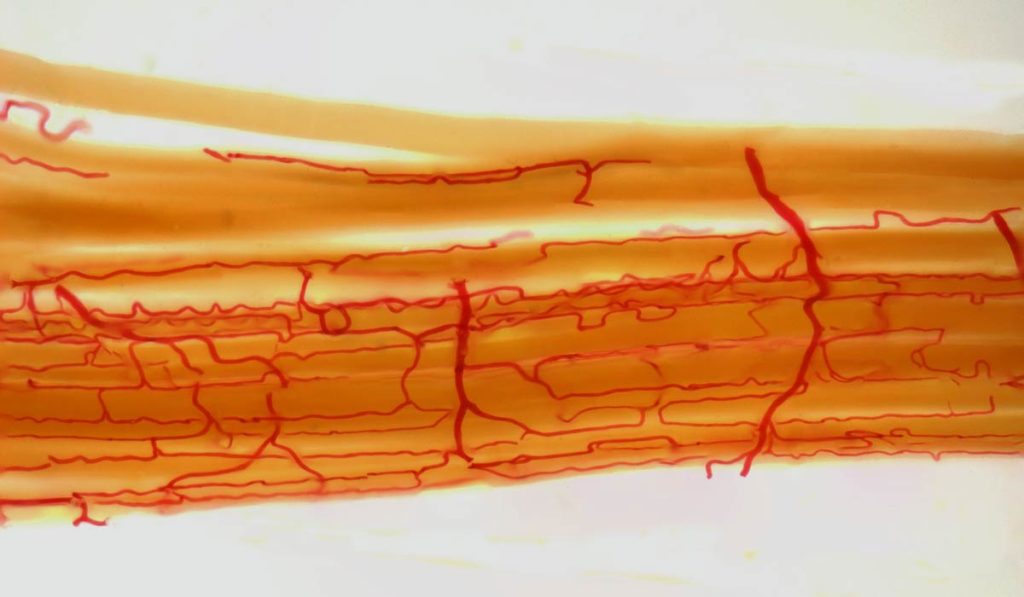
Microvascular disease (MVD) plays a large part in raising amputation risk. This role is independent of peripheral artery disease (PAD), though in combination the risks are elevated further. Until now, a lack of data on the role of MVD has meant that clinicians may focus almost entirely on PAD and diabetes as potential precursors to non-traumatic limb loss.
A new study published in the journal Circulation may change that perspective. Research led by Joshua Beckman, M.D., director of vascular medicine at Vanderbilt University Medical Center, examined the hospital records of 125,674 veterans, including 1,185 patients who had lower limb amputations. Using billing codes for retinopathy, nephropathy and neuropathy, they identified veterans with PAD and MVD.
MVD alone was associated with a 3.7-fold increased risk for amputation and was the cause of 18 percent of all the amputations. PAD alone elevated risk 13.9-fold, and the combination of MVD and PAD resulted in a 22.7-fold higher risk.
“The presence of MVD helps identify a population not previously considered at particularly high risk for amputation.”
“This research shows that the presence of MVD helps identify a population not previously considered at particularly high risk for amputation,” Beckman said. The study also showed that MVD anywhere in the body augurs systemic MVD. “If a patient has retinopathy or nephropathy, it’s a signal that they are likely to have MVD elsewhere as well,” he said.
Impact of MVD
Large vessel disease and MVD are related, but the overlap is not complete. Disease conditions that exist outside the overlap can lead to significantly different outcomes. For example, patients who have had angioplasty and stenting after myocardial infarction may not experience symptoms of cardiovascular disease if they do not also have MVD.
Patients with MVD whose capillaries and small arterioles have “dropped out” of circulation typically do not heal as well after leg bypass surgery compared to patients with PAD. They are more prone to pain while walking and suffer from sores on their feet that have difficulty healing regardless of the blood flow in the larger vessels. “Our study provides clinical evidence that MVD diagnosed in any vascular bed likely increases the risk of dermal microvascular dysfunction, poor wound healing and amputation,” Beckman said.
While this may make intuitive sense, Beckman says the condition of microvessels tends to be overlooked in this setting. “Now, we have evidence that MVD does not necessarily proceed hand-in-hand with other cardiovascular diseases, that its presence along with PAD substantially amplifies risk, and that isolated MVD represents a systemic warning,” Beckman said. “It would make sense for clinicians to adjust testing, therapy and prognoses to incorporate this understanding.”
Diabetes: an Independent Risk Factor
While diabetes contributes to MVD and PAD, the data showed a similar increase in risk of amputation in patients with and without diabetes. “Based on the data, it appears diabetes is another mechanism that would make limb problems more likely,” Beckman said.
In the subgroup of patients with MVD, diabetics were in the minority, underscoring that MVD is a significant risk factor independent of diabetes. In light of these findings, Beckman thinks clinicians may do well to shift from associating retinopathy and other MVD manifestations only with diabetes and increase their vigilance with symptomatic non-diabetics, such as those with hypertension.
The study is one arm of a multicenter American Heart Association (AHA) Strategically-focused Research Network grant, for which Beckman serves as coordinator for the Vanderbilt site. The grant is also funding research to explore how muscle metabolism, microvascular architecture and genetic profiles contribute to PAD risk.