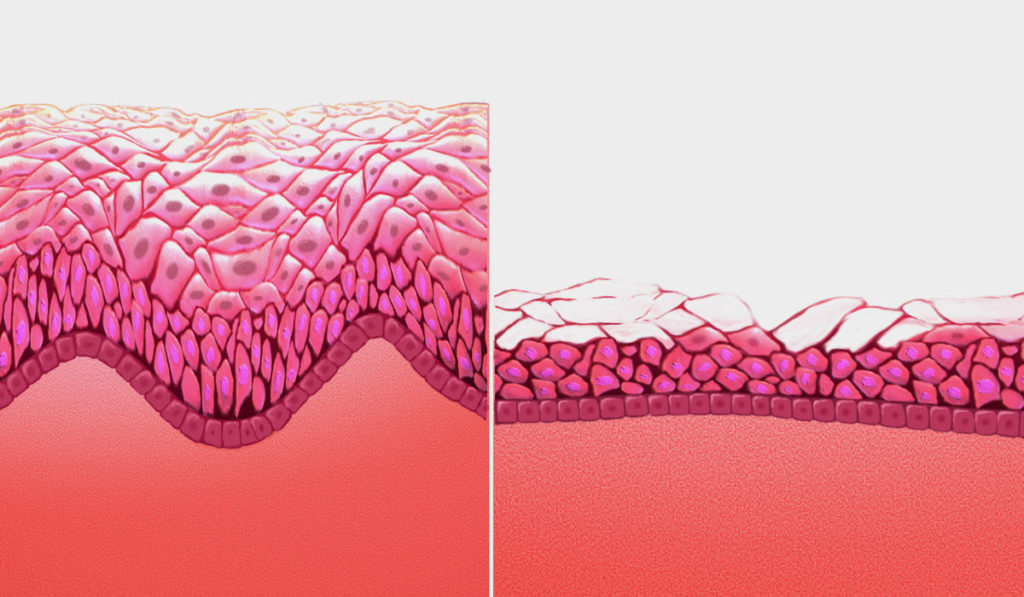
Genitourinary syndrome of menopause (GSM) describes a variety of menopausal symptoms including genital (dryness, burning and irritation), sexual (lack of lubrication, discomfort or pain, and impaired function) and urinary symptoms (urgency, dysuria, and recurrent urinary tract infections). GSM can be debilitating and can profoundly impact quality of life. It is thought to affect about half of postmenopausal women.
A recent retrospective study found microablative fractional carbon dioxide (CO2) laser therapy could improve GSM management when a series of laser treatments is applied in a follow-up period of 12 months. A new clinical trial, sponsored by the FDA and the manufacturer of the MonaLisa Touch® laser, will follow patients with GSM undergoing the treatment. The trial will begin recruiting in late summer-early fall; Vanderbilt University Medical Center is one of 10 study sites.
“When women lose estrogen through menopause or surgery, vaginal dryness is a very prominent symptom.”
“When women lose estrogen through menopause or surgery, vaginal dryness is a very prominent symptom,” said Melinda New, M.D., associate professor of clinical obstetrics and gynecology and co-founder of the Vanderbilt Center for Vulvar and Vaginal Disorders. “Many of our patients are breast cancer survivors who do not want to take estrogen. And for some patients, many of the over-the-counter remedies either don’t work or the women don’t want to risk worsening their menopausal side effects.”
The GSM Study
The MonaLisa Touch®, designed and produced by DEKA (Italy), delivers fractional CO2 laser energy to vaginal mucosa, stimulating collagen production and triggering a healing response. “The skin appears healthier and more resilient: better collagen and elastin, increased blood flow, and more layers,” New said.
The trial—a multi-center, randomized, blinded, crossover trial looking at treatment of vaginal atrophy only —will be the first of its kind using the CO2 laser device. New and Lisa Milam, M.S.N., an instructor of clinical obstetrics and gynecology at Vanderbilt, have been using the device off label to treat vaginal atrophy since March 2018. Their typical protocol is three treatments, spaced six weeks apart.
“We’ve been very academic and rigorous in our approach to treating this disorder and we’ve had great anecdotal success,” New said. “In 15 months, not one patient has come back needing additional treatment. We’re hoping this study will open the door for FDA approval and for more supportive clinical studies.”
Beyond GSM
New and Milam have been using the device to treat other vulvar and vaginal disorders, including dermatologic issues such as lichen sclerosis. Most of these disorders are heterogeneous in appearance, and it can sometimes take years for women to be correctly diagnosed.
“Many patients have experienced symptoms for years, have seen multiple specialists, and tried various treatment options without success.”
“These chronic disorders can be really frustrating, sometimes debilitating, and can dramatically affect a postmenopausal woman’s quality of life and sexual relationships,” Milam said. “Many patients have experienced symptoms for years, have seen multiple specialists, and tried various treatment options without success.”
In treating vulvovaginal dermatologic disorders, the team assesses each patient holistically and develops a personalized treatment approach. They have found that patients with hypertrophic changes such as thickened skin show better response than those with thinner, excoriated skin. More research needs to be done to develop treatment protocols and evidence-based therapies, New said.
“As a community, we still don’t know much about these disorders,” she added. “It would be an important adjunct to what we can offer women in our clinic to provide a therapeutic answer to these usually severe problems.”